JDRF Blog
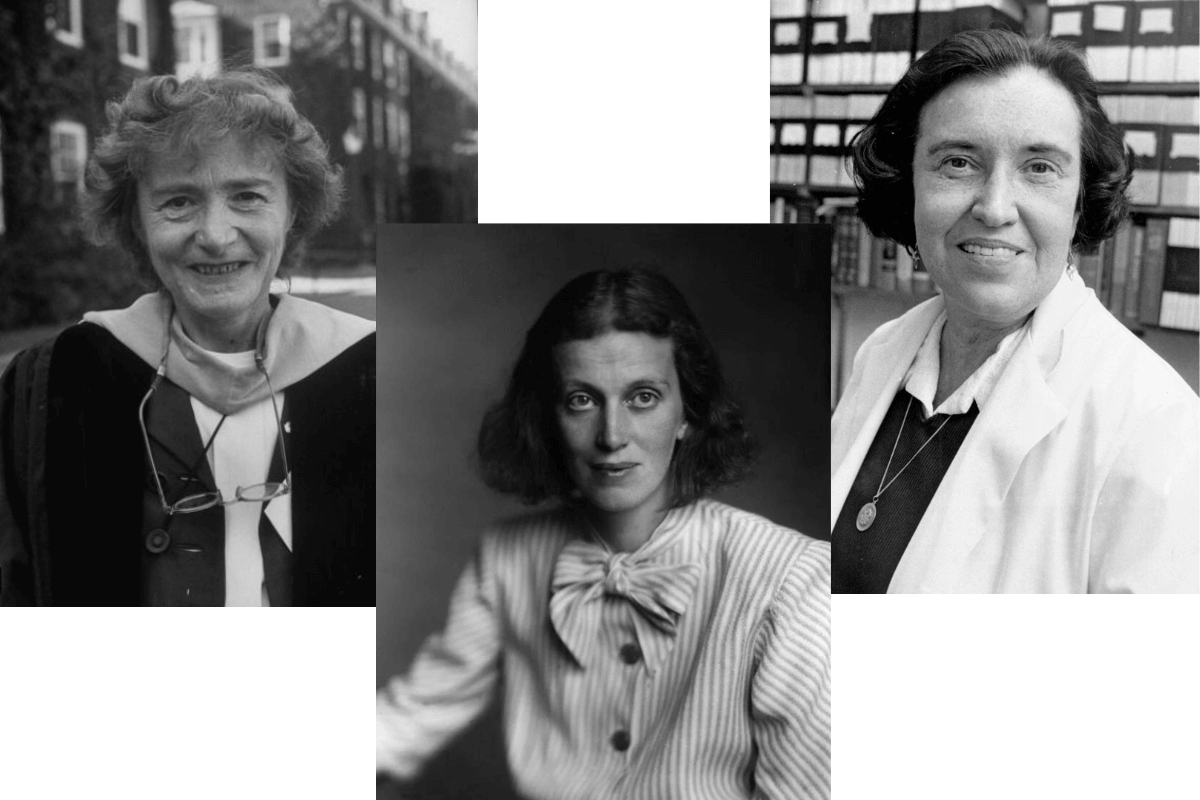
Nobel Laureates, Women, and Diabetes Discoveries
Gerty Cori, Ph.D., Dorothy Crowfoot Hodgkin, FRS, and Rosalyn Yalow, Ph.D., won the Nobel Prize for diabetes discoveries.
Read More About Nobel Laureates, Women, and Diabetes Discoveries
ATTD Brings Together Top Minds in Diabetes Research
Leading researchers gathered for the ATTD meeting, which featured more than 50 studies presented by JDRF-funded researchers.
Read More About ATTD Brings Together Top Minds in Diabetes Research
We are Grateful to be a Part of the T1D Community
In 2023, we have made tremendous progress in accelerating cures, improving lives, and advocating for people with T1D and their loved ones. Take a look.
Read More About We are Grateful to be a Part of the T1D Community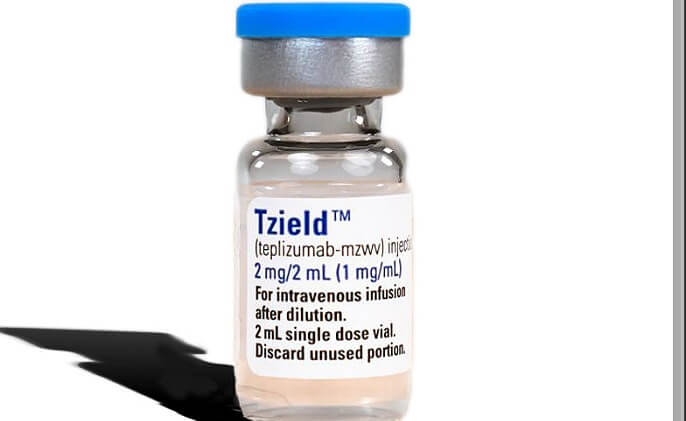
Results Are Out: Tzield Benefits Newly Diagnosed Individuals
A new study published in the New England Journal of Medicine shows Tzield can preserve beta cell health in certain individuals recently diagnosed with T1D.
Read More About Results Are Out: Tzield Benefits Newly Diagnosed IndividualsRecent Articles
Sign up for our monthly Research Newsletter, The Pipeline, to stay up to date on the latest and greatest in T1D science.
By clicking Sign Up, I agree to the JDRF Privacy Policy. I also agree to receive emails from JDRF and I understand that I may opt out of JDRF subscriptions at any time.